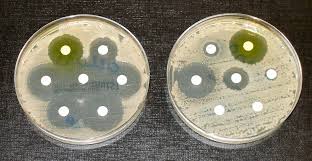
We’ve all heard the antibiotic resistance horror stories: superbugs are going to take over the world; antibiotics are going to stop working, leaving the human race trampled by bacteria that we can do nothing to kill. There were 450,000 new cases of multi-drug resistance tuberculosis in 2012 (according to the World Health Organisation) – and that’s just the tip of the antibiotic resistant iceberg.
Antibiotic resistance is the ability for a bacterium to survive when exposed to a certain antibiotic: the bacterium is no longer sensitive to that particular antibiotic and so can carry on dividing, and making us feel ill.
Antibiotic resistance genes are sections of DNA that encode for proteins which damage bacterial cells in some way. Ways they do this include:
- Breaking down antibiotics
- Removing antibiotics from cells
- Replacing the enzyme that is broken down by the antibiotic with another which can do the same job, but is not targeted by the antibiotic
- Making the bacterial cell less permeable, so antibiotics can’t get into the cell (TB does this, which is what can make it so difficult to treat)
Antibiotic resistance can be transmitted vertically, which means that when a bacterial cell divides, the gene is passed on to its offspring, or horizontally, which means it is transferred via a plasmid (a small, circular loop of DNA), to a different bacterial cell. The process of horizontal transfer is called bacterial conjugation, when a bridge-like connection is made between two bacterial cells, and plasmid DNA is copied and a plasmid transferred into another cell, taking the antibiotic resistance gene with it.
Antibiotic resistance didn’t spring up all of a sudden when we started using antibiotics; it already existed in bacteria when Flemming discovered penicillin, though they were not particularly effective at first and have since become more common with growing antibiotic use. Bacteria divide every 18-20 minutes, so increase by tenfold every hour, allowing them to adapt to environmental changes far quicker than we can. When a selection pressure (some form of stress which prevents all bacteria from surviving) is applied, for example, the presence of an antibiotic, those which are vulnerable to the antibiotic die, while those which aren’t (those with a gene allowing resistance to the antibiotic) survive, and go on to reproduce, ending up with a much greater proportion of the surviving bacteria possessing the antibiotic resistance gene.

Resistance is a huge problem which we may be yet to see the head of. While research is going into developing new antibiotics and ways of getting around the antibiotic resistance issue, potentially by inhibiting enzymes which break down antibiotics, there’s also a part we have to play to lessen the spread of antibiotic resistance. Doctors often prescribe antibiotics when they’re unsure if the infection is bacterial or viral (if it is viral, antibiotics will have no effect), but it might not, and sometimes even when they might not even be entirely necessary and the infection might clear up in a couple of days on its own. Antibiotics can also harm the “good” bacteria in our body, including our digestive system, which is why some people have trouble digesting certain foods, particularly dairy, for a little while after a course of antibiotics. We need to be a little more selective when deciding when to take antibiotics (and doctors need to be more selective about prescribing them), and when we do take antibiotics, it’s important to finish the entire prescribed course, to prevent the infection returning and becoming resistant.
How do you think the prescription of antibiotics should be controlled? Add your comments and ideas by clicking the comments button at the top of this post.

Hi, lovely blog post, really well written especially for an undergrad!
Although the doubling time for E. coli can be just 20 minutes, this is one of the quickest bacterial doubling times. M. tuberculosis for example can take over anywhere from 14 to over 24 hours according to some quick reading! http://www.ncbi.nlm.nih.gov/pubmed/10792526
LikeLike
Thank you for your comment! That’s really interesting, especially as while that sounds like much longer, in terms of generation times in general, that’s still incredibly short compared to most other organisms.
LikeLiked by 1 person
It’s ok! Well in microbial terms it is a lot longer, but yeah compared to eukaryotes it is still scarily quick!
LikeLike
Yeah you’re right, that is a huge difference isn’t it? That really surprised me actually; I didn’t realise they could be that long!
LikeLiked by 1 person
If you take it down to the microbial timescale, it is huge! Imagine 2-3 multiplications of E. coli per hour (48-72 x!), vs just 1 multiplication of M. tuberculosis in 24 hours! Yeah and then some extremophiles can be really slow growing. I know methanotrophs ans cyanobacteria are really slow growers, can take weeks for colonies to appear on agar, as opposed to overnight with E. coli.
LikeLike
Thank you. The recent superbug outbreak in India is very telling. Unfortunately, the world is still captured by Ebola and its victims in West Africa and elsewhere.
LikeLike